11 | Add to Reading ListSource URL: www.sp.seLanguage: English - Date: 2013-10-10 04:16:46
|
|---|
12 | Add to Reading ListSource URL: wtgevents.comLanguage: English - Date: 2014-02-12 07:28:27
|
|---|
13 | Add to Reading ListSource URL: www.pulmatrix.comLanguage: English - Date: 2012-09-04 07:00:22
|
|---|
14 | Add to Reading ListSource URL: www.ijaponline.orgLanguage: English - Date: 2011-05-29 06:46:22
|
|---|
15 | Add to Reading ListSource URL: www.pulmatrix.comLanguage: English - Date: 2014-12-11 12:27:38
|
|---|
16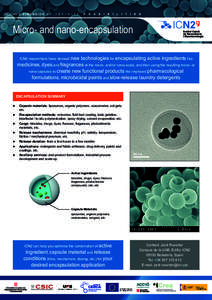 | Add to Reading ListSource URL: www.icn.catLanguage: English - Date: 2013-10-29 10:16:39
|
|---|
17 | Add to Reading ListSource URL: ajpcr.comLanguage: English - Date: 2014-07-10 04:13:44
|
|---|
18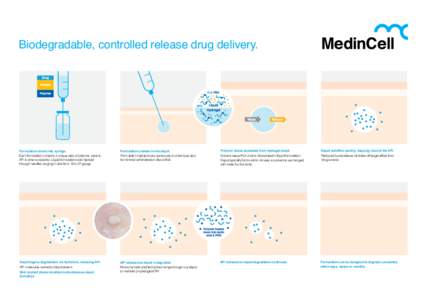 | Add to Reading ListSource URL: www.medincell.euLanguage: English - Date: 2015-02-01 13:11:30
|
|---|
19 | Add to Reading ListSource URL: www.ijaponline.orgLanguage: English - Date: 2011-05-29 06:46:18
|
|---|
20 | Add to Reading ListSource URL: 216.194.172.131Language: English - Date: 2013-11-07 17:12:17
|
|---|